Tag you’re it…Supplier Quality
The childhood game of “tag” may be harmless fun, but in product development it often takes a far less productive form: shifting responsibility to vendors and suppliers. Product developers frequently hear explanations such as, “the vendor did not process the document,” or “the supplier failed to consider regional or regulatory requirements.” In most cases, these issues point not to supplier failure, but to gaps in the company’s Supplier Quality program.
For medical device and pharmaceutical product developers, Supplier Quality is a critical enabler of execution, whether working with Contract Manufacturing and Distribution Organizations (CMDOs), Contract Research Organizations (CROs), or specialized consultants. While many organizations maintain robust Supplier Quality controls for GMP manufacturing, the same rigor is often missing for development and service-based activities. Legal agreements may exist, covering areas such as distribution, clinical services, or pharmacovigilance, but without clearly defined expectations, deliverables, and accountability, risk remains unmanaged.
An effective Supplier Quality program must be integrated into the new product development process, not added after issues arise. Supplier qualification and oversight should evolve across the entire product lifecycle, from concept and development through commercialization and post-market support. This requires deliberate cross-functional handoffs, aligned responsibilities, and phase-appropriate controls. Early identification and qualification of suppliers accelerates timelines by leveraging external expertise, while thoughtful selection based on long-term needs improves cost transparency and supports smooth market launches.
(See: https://consultwing.com/product-development-tplc)
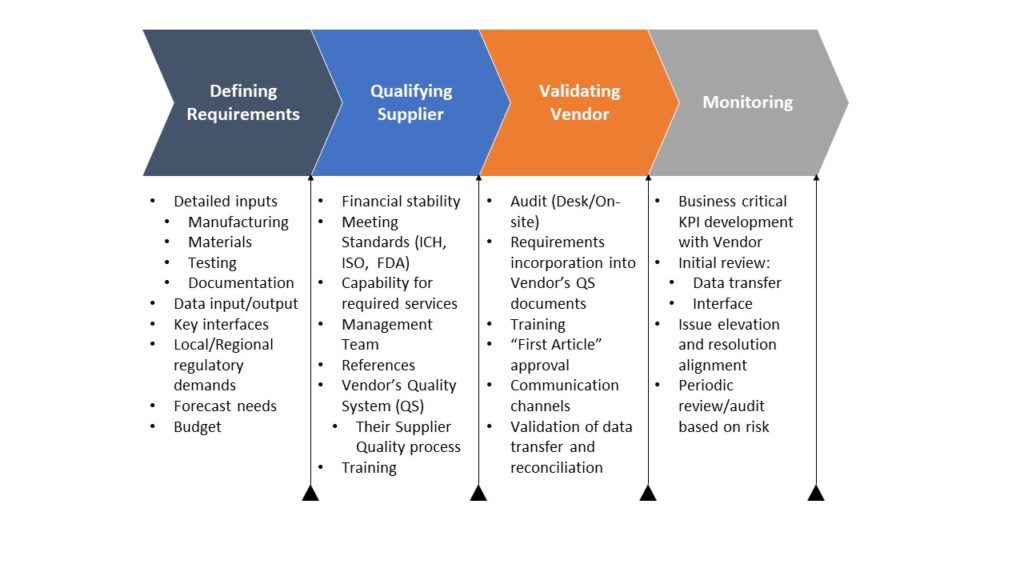
Think of suppliers as products. Define requirements, expected outputs, and measurable KPIs. Establish auditing and performance monitoring that drives continuous improvement. This approach minimizes finger-pointing, strengthens partnerships, and maximizes supplier value.
Consult Wing supports organizations in integrating Supplier Quality into end-to-end Product Lifecycle Management, including:
- Project design: Clear deliverables, decision rights, and communication pathways
- Risk management: Patient- and product-focused risk assessment and decision-making
- Quality system infrastructure: Scalable Supplier Quality frameworks aligned to development and commercialization
We help product developers align, achieve, and sustain business-driven quality processes that enable speed, compliance, and patient safety.
