Posts by Consult Wing
From Checklists to Common Sense:How ICH GCP E6(R3) and QSMR Finally Made Quality Grow Up
For years, “quality” in clinical research and regulated product development has had an identity problem. It was often confused with: If it felt like quality was something you survived rather than used, you weren’t wrong. Enter ICH GCP E6(R3) and the FDA’s Quality System Management Review (QSMR), two frameworks that quietly but decisively change the…
Read More2025 FDA 483 Review: Why Combination Products Are Disproportionately at Risk
Combination products sit at the intersection of drug CGMPs, device QSR/QSMR, and Part 4 integration requirements. FDA 483 data across both sectors show that organizations continue to manage these requirements in parallel rather than within an integrated quality system, a structural weakness that QSMR will further expose. MEDICAL DEVICES:2025 FDA 483 OBSERVATIONS DESCRIPTION Frequency Lack…
Read MoreFrom Exception to Action: Risk-Based Issue Elevation
Under FDA’s Quality Management System Regulation (QMSR), exception handling is no longer about closing deviations—it is about demonstrating risk-based thinking across the product lifecycle. This expectation applies equally to pharmaceuticals, medical devices, and combination products, where disconnected investigations often delay CAPA or trigger avoidable field actions. The differentiator is how exceptions are understood and elevated.…
Read MoreNot All Combination Products Are Created Equal: The Prefilled Syringe (PFS) Challenge
Prefilled syringes may seem straightforward: select a syringe, fill it with drug product, and contract out manufacturing, but in reality, PFS development is a complex endeavor. Regulatory agencies like the FDA are placing increased scrutiny on PFS-related combination products, citing a growing number of compliance issues related to design, testing, and quality systems. The Complexity…
Read MorePodcast on Thriving in the Medical Device Industry: Quality and Regulatory Organisation and processes
Plan Now Pays Later
Typically, initial planning neglects to account for potential failures in manufacturing, clinical testing, or handling post-marketing complaints. This oversight can lead to significant setbacks. Prioritizing planning for success over acknowledging potential setbacks isn’t the most optimistic approach; instead, understanding the deliverables across the organization is paramount. While regulations often lag behind, organizations prioritizing business-centric processes…
Read MoreIoT presentation on Telemedicine
Developing Quality Relationships
For years, I have been mentoring a successful Quality/Regulatory friend who recently asked for guidance on dealing with personal relationships, focusing on his career and personal relationships. We discussed how we have established goals and requirements in our business lives that we often overlook in our personal lives. Having mentored him professionally, my guidance was…
Read MoreWhat is Essential is Invisible to the Eye
For Quality Executives, communication is not simply about information transfer; it is about insight, context, and judgment. One of the enduring lessons from The Little Prince captures this well: “It is only with the heart that one can see clearly; what is essential is invisible to the eye.” That message is particularly relevant in regulated…
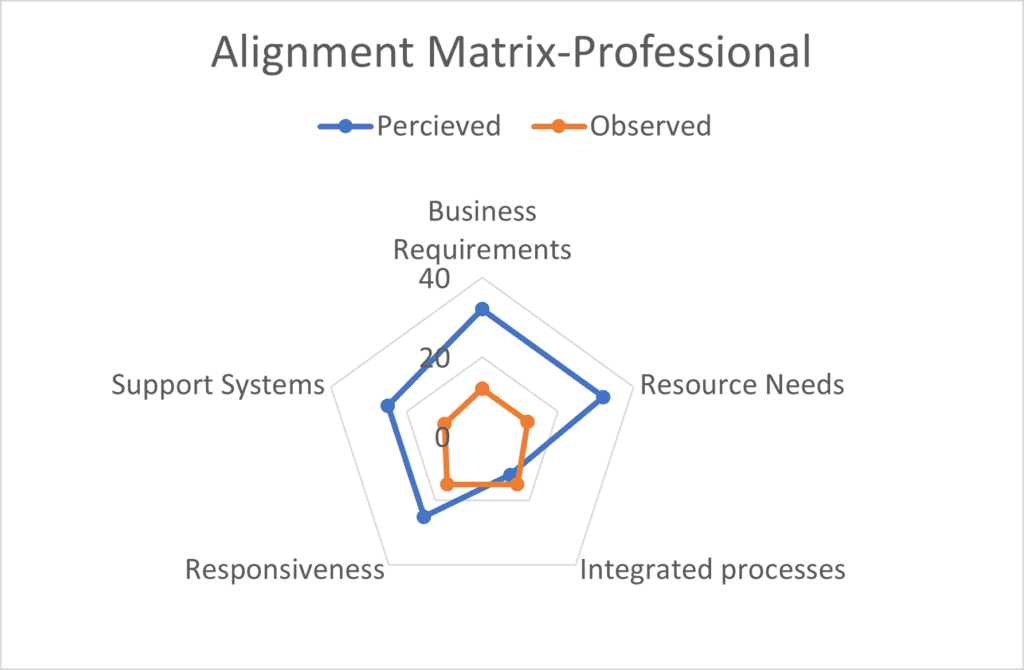
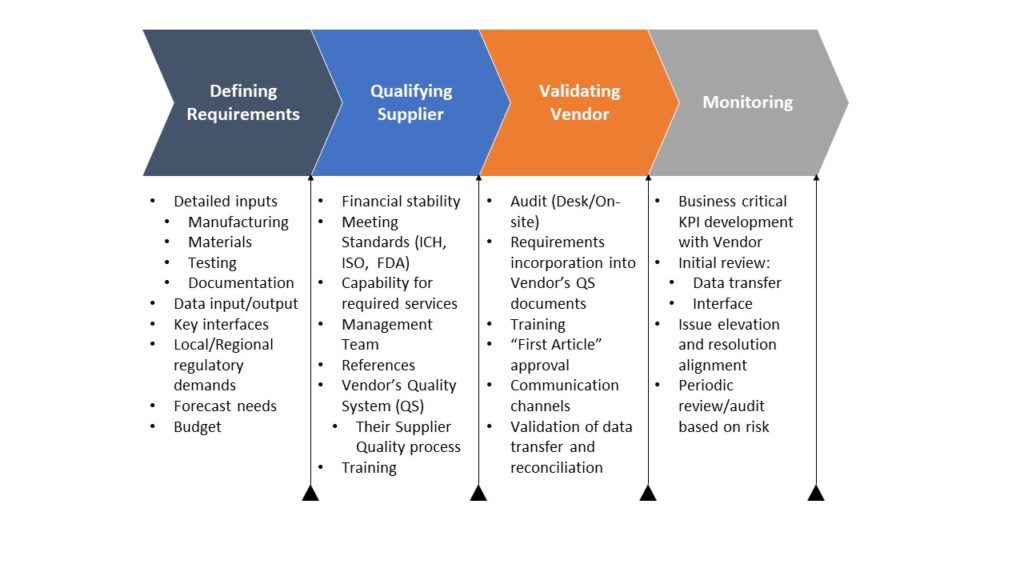
Read MoreTag you’re it…Supplier Quality
The childhood game of “tag” may be harmless fun, but in product development it often takes a far less productive form: shifting responsibility to vendors and suppliers. Product developers frequently hear explanations such as, “the vendor did not process the document,” or “the supplier failed to consider regional or regulatory requirements.” In most cases, these…
Read More