Striving for perfection
For Quality Executives, one of the most consequential leadership challenges is knowing when to pursue improvement and when to recognize the point of diminishing returns. A lesson I first encountered in a high school geometry class captures this well. While discussing hyperbolic equations, the teacher paused and observed, “Man is like a hyperbola, always striving toward the asymptote.” Its meaning became clear only years later.
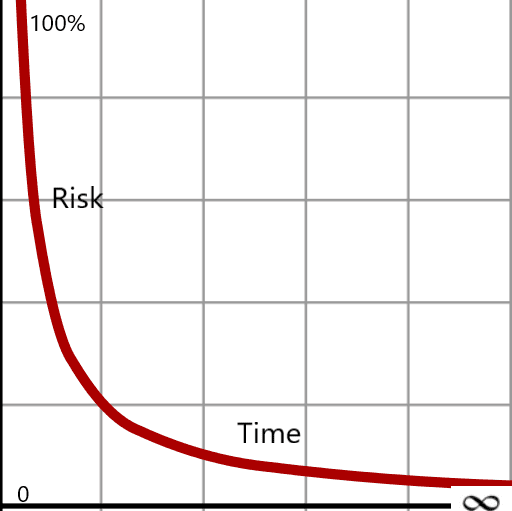
In a hyperbola, the curve approaches the asymptote indefinitely but never intersects it. The closer it gets, the more effort is required for increasingly marginal progress. This mirrors a common pitfall in quality and regulatory work: the pursuit of a “perfect” outcome.
Just as the Great Buddha stated, the harder you search for something, the further away it gets. Focusing on a perfect outcome, just like a hyperbola, can consume time and your sanity.
In regulated product and process development, perfection, defined as zero risk, is not achievable. Excessive focus on eliminating every residual risk can consume time, strain resources, and delay patient benefit without meaningfully improving outcomes. This is where disciplined risk management becomes essential.
Risk management acknowledges a fundamental truth: zero risk does not exist. Risks can be identified, evaluated, controlled, and monitored, but uncontrollable variables, human factors, process variability, material tolerances, and operational complexity will always remain. The role of quality leadership is not to eliminate all risk, but to understand it, prioritize it, and determine where mitigation efforts meaningfully reduce the likelihood or severity of catastrophic failure.
The organizational challenge is aligning risk tolerance with available resources and timelines across the product lifecycle, whether the risk relates to patient safety, manufacturing robustness, validation strategy, or Quality System controls. For every activity, leaders must make deliberate decisions about the acceptable level of residual risk and where additional investment yields diminishing returns.
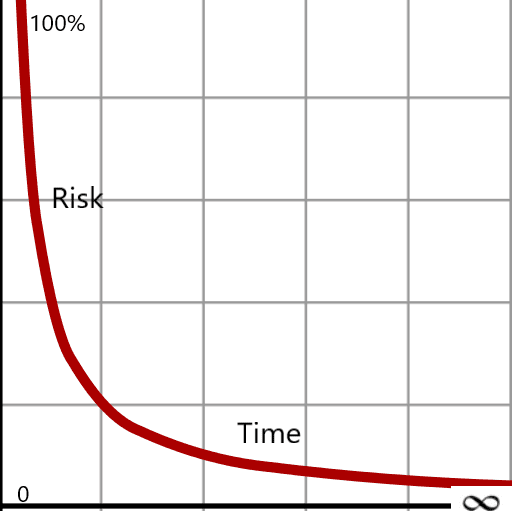
Risk management should be the foundational tool at the start of any initiative, new drug development, device design, or process implementation, establishing a shared understanding of risk tolerance before project plans are finalized. While we may strive for minimal risk, an infinite investment of time and effort will not guarantee a risk-free outcome.
Does your organization consistently integrate risk management into product and process development decisions? Consult Wing helps Quality Executives apply pragmatic, lifecycle-based risk management to balance compliance, speed, and patient safety. We bring the right tools and perspective to help you manage risk intelligently across the enterprise.
In simple terms, risk management helps you understand how wide the stream truly is, so you know when, where, and how to jump safely to the other side.
