Tag you’re it…Supplier Quality
The childhood game of ‘tag’ is fun, I still enjoying playing with young children. However, in business the game of ‘tag’ is often used as an excuse for pushing responsibilities on to vendors/suppliers. How many times have you heard ‘the vendor didn’t process a document’ or the ‘supplier forgot to consider local or regional requirements?’ Your Supplier Quality program is the key to success whether using a Contract Manufacturer and Distribution Organization (CMDO) or Contract Research Organization (CRO) or even hiring a consultant.
Companies may have a strong Supplier Quality process for manufacturing (GMP) processes. Others might have legal agreements to cover services, ranging from distribution to clinical and post-market safety (PharmacoVigilance Agreements-PVA) without truly defining/understanding what they want/need from the supplier. Do you integrate your Supplier Quality program into your new product development process?
Vendor qualification evolves throughout the product life-cycle management, starting at concept through Post-market support. Requiring cross-functional and intra-functional handoffs through the different phases of the project (See https://consultwing.com/product-development-tplc). Identifying and qualifying suppliers helps expedite the timelines utilizing their resources and expertise. Choosing wisely with long-term needs provides organizations detailed understanding of development cost and transitional planning for successful market launches.
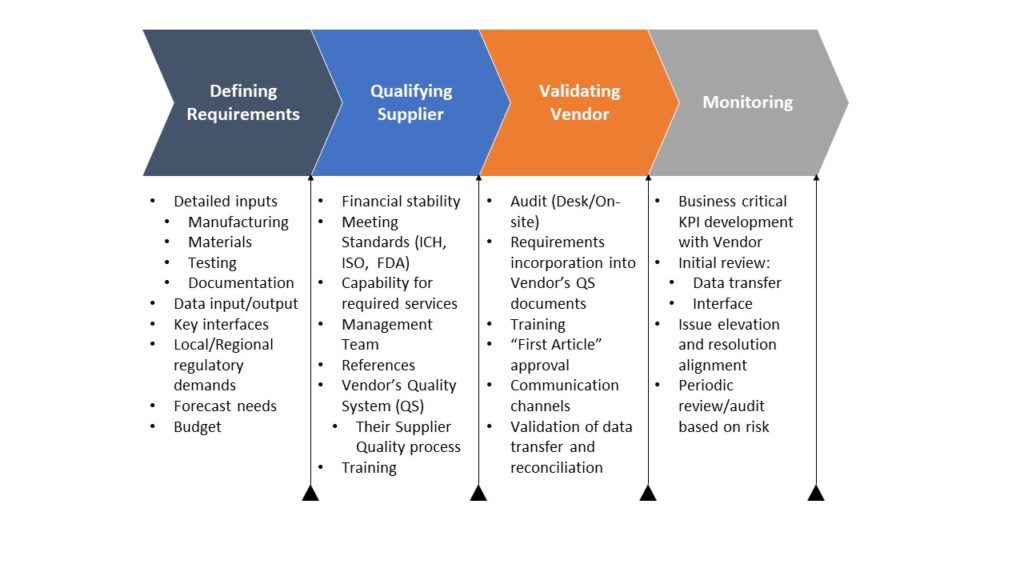
Global Supplier Quality Program
Treat your suppliers as a new product; defining requirements, outputs, along with measurable KPIs for improvements and auditing will minimize finger-pointing and enhance the utilization of your suppliers.
Consult Wing has helped organizations through the integration of all aspects of their Product Life Cycle Management:
- Project design; focusing on deliverables, decision rights and communication
- Risk Management; focusing on patient and product risk determination and decision-making
- Quality System infrastructure support, including Supplier Quality
Let us help you align, achieve, and sustain your business and quality processes.
